Product Enquiry
If you would like to send us an enquiry about this product, please click the button below, fill in the form and submit.
Product EnquiryPreciseInhale®
Inhaler Aerosol Generator
Because the aerosol produced by PreciseInhale® is controlled, it is easily exposed in a range of exposure modules. The aerosol can be sourced from dry powders, inhalers or nebulized solutions, optimizing conditions for a wide range of substrates. PreciseInhale® works as an exposure platform for small-scale inhalation experiments by precisely dosing animals in vivo, lungs ex vivo, and depositing material for in vitro exposure and dissolution testing.
Separating aerosol generation and dispensingAerosols are generated with a high-power air jet of 10-160 bar pressure. The energy is then siphoned off, leaving a plume of aerosol that is ‘pulled’ into a fine stream by a vacuum pump, enabling it to be dispensed separately across a range of exposure modules. |
PreciseInhale®’s software monitors and measures aerosol in real time.Automated software and a light-scattering device maps and measures the aerosol concentration and breathing pattern in real time. This enables researchers to customize their aerosol to their exact needs, and generates detailed voluminous PK data, including particle size distribution data and Tmax and Cmax curves. |
In vivo Nose-Only Exposure |
 |
This nose-only module exposes single rodents to respirable aerosols from PreciseInhale®. Safely, gently, and precisely, reducing standard deviation, the number of test animals needed, and obtaining highly reliable pharmacokinetic data. |
|
In vivo Intratracheal Exposure |
 |
With the module for intratracheal inhalation exposures of rodents to respirable aerosols by the PreciseInhale® system, it is possible following administration to obtain pharmacokinetic data resembling clinical inhalation exposures. |
|
In vitro Dissolution and Absorption |
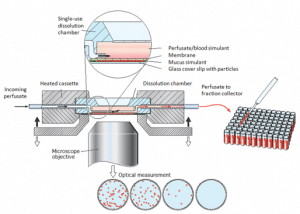 |
DissolvIt ® enables in vitro simulation of absorption and dissolution of respirable particles in the lung epithelium.
|
In the patented DissolvIt®, in vitro module, dry powder particles are deposited on a glass surface by PreciseInhale®. The particles are then brought into contact with a simulated lung/airway mucus and the dissolution is studied, from the ‘luminal’ side through optical microscopy and from the ‘vascular’ side by chemical analysis of a flow-past perfusion medium. |
In vitro Cell Exposure |
 |
XposeALI ®3D cell exposure module combines aerosol capability with 3D cell models cultured at an Air-Liquid Interface (ALI). It enables studies of cellular effects induced by airborne particles. |
|
Features
- <100 mg of substance for a complete PK-study
- Precision dosing, <10% SD
- Flexible aerosol sources: Dry Powder (API) Nebulized solution Inhalers (DPI/ pMDI)
- IVIVC – minimized translational errors
- 3R –reduce, refine and replace animal tests
Research Areas
- Asthma
- Influenza
- Diabetes
- Migraine
- Chronic obstructive pulmonary disease
Applications
- Systemic pharmacokinetics
- Lung specific pharmacokinetics
- Early toxicology testing
- Dissolution and absortion testing
- Ranking of drug candidates
- Vaccine development
- Aerosol particle size determination
- Toxicology studies
- Nanoparticle exposure pollutants, e.g. diesel soot exposure
- Cellular effects after exposure
Selected Publications
Per Gerde, Pär Ewing, Lena Låstbom, Åke Ryrfeldt, Juri Waher & Göran Lidén (2004) A Novel Method to Aerosolize Powder for Short Inhalation Exposures at High Concentrations: Isolated Rat Lungs Exposed to Respirable Diesel Soot, Inhalation Toxicology, 16:1, 45-52, DOI: 10.1080/08958370490258381
Alessandro Fioni, PhD, Ewa Selg, MD, PhD, Valentina Cenacchi, BS, Fernando Acevedo, PhD, Giandomenico Brogin, BS, Per Gerde, PhD, and Paola Puccini,Investigation of Lung Pharmacokinetic of the Novel PDE4 Inhibitor CHF6001 in Preclinical Models: Evaluation of the PreciseInhale Technology, JOURNAL OF AEROSOL MEDICINE AND PULMONARY DRUG DELIVERY. Volume 30, Number 0, 2017, Pp. 1–10, DOI: 10.1089/jamp.2017.1369
Per Gerde, Maria Malmlof, Lina Havsborn, Carl-Olof Sjöberg, Pär Ewing, Stefan Eirefelt, and Katarina Ekelund 5,* DissolvIt: An In Vitro Method for Simulating the Dissolution and Absorption of Inhaled Dry Powder Drugs in the Lungs ASSAY and Drug Development Technologies, VOL. 15 NO. 2 FEBRUARY/MARCH 2017 DOI: 10.1089/adt.2017.779
Ji J, Hedelin A, MalmloÈf M, Kessler V,Seisenbaeva G, Gerde P, et al. (2017) Development of Combining of Human Bronchial Mucosa Models with XposeALI® for Exposure of Air Pollution Nanoparticles. PLoS ONE 12(1): e0170428. doi:10.1371/journal.pone.0170428
Per Gerde, PhD, Mattias Nowenwik, MSc, Carl-Olof Sjöberg, MSc, and Ewa Selg, MD, PhD Adapting the Aerogen Mesh Nebulizer for Dried Aerosol Exposures Using the PreciseInhale Platform JOURNAL OF AEROSOL MEDICINE AND PULMONARY DRUG DELIVERY. Volume 32, Number 0, 2019, Mary Ann Liebert, Inc. Pp. 1–11, DOI: 10.1089/jamp.2019.1554
Selected Posters
Biorelevant dissolution to differentiate formulation performance for inhalation. Beatriz Noriega (Hovione FarmaCiencia SA, Portugal /Med.ULisboa, Faculdade de Farmácia, Universidade de Lisboa) Maria Malmlöf, Per Gerde (Inhalation Sciences, Huddinge, Sweden / Institute of Environmental Medicine, Karolinska Institutet, Sweden) Mattias Nowenwik (ISAB) Luisa Corvo2 , Eunice Costa (Med.ULisboa, Faculdade de Farmácia, Universidade de Lisboa) Poster 58, DDL 2019 Conference, December 2019.
Characterisation of aerosols from solution-based pressurised metered dose inhalers (pMDIs) when delivered by PreciseInhale®. Wachirun Terakosolphan (Institute of Pharmaceutical Science, King’s College London, UK), Maria Malmlöf (ISAB, Sweden), David Lewis (Chiesi Limited, Chippenham, UK), Ben Forbes (Institute of Pharmaceutical Science, King’s College London, UK). DDL poster 2017.
Aerosolizing PulmoSolTM and PulmoSphereTM with the PreciseInhale® system. Nowenwik, M (ISAB, Sweden), Malmlöf, M (ISAB, Sweden), Kadrichu, N (Novartis USA), Gerde, P (ISAB, KI Institute of Environmental Medicine, Sweden). ISAM conference poster, 2017.
Effect of Compressed Air Pressure on Particle Size Distribution During Aerosol Generation with Ultrafine TiO2. Maria Malmlöf (ISAB), Mattias Nowenwik (ISAB), Per Gerde (ISAB, KI Institute of Environmental Medicine), DDL poster 2017.
Effect of using bio-relevant media in the DissolvIt ® system to measure dissolution of fluticasone propionate from Flixotide 50 µg Evohaler, joint poster King’s College London, ISAB. Authors: Mireille Hassoun, Maria Malmlöf, Abhinav Kumar, Sukhi Bansal, Mattias Nowenwik, Per Gerde, Mark Parry, Ben Forbes. Poster: DDL27, 2016.
Selected Product Citations
Dry Generation of CeO2 Nanoparticles and Deposition onto a Co-Culture of A549 and THP-1 Cells in Air-Liquid Interface—Dosimetry Considerations and Comparison to Submerged Exposure |
|
| Francesca Cappellini, Sebastiano Di Bucchianico, …, Hanna L Karlsson | Nanomaterials Basel | Published 27 Mar 2020 |
| Article Snippet Abstract: Relevant in vitro assays that can simulate exposure to nanoparticles (NPs) via inhalation are urgently needed. Presently, the most common method employed is to expose lung cells under submerged conditions, but the cellular responses to NPs under such conditions might differ from those observed at the more physiological air-liquid interface (ALI). The aim of this study was to investigate the cytotoxic and inflammatory potential of CeO2 NPs (NM-212) in a co-culture of A549 lung epithelial cells and differentiated THP-1 cells in both ALI and submerged conditions. Cellular dose was examined quantitatively using inductively coupled plasma mass spectrometry (ICP-MS). The role of serum and LPS-priming for IL-1β release was further tested in THP-1 cells in submerged exposure |
|
Use of PBPK Modeling To Evaluate the Performance of DissolvIt, a Biorelevant Dissolution Assay for Orally Inhaled Drug Products |
|
| Mol Pharm | Published 14 Jan 2019 | |
| Article Snippet Abstract: Open in a separate window The dissolution of inhaled drug particles in the lungs is a challenge to model using biorelevant methods in terms of (i) collecting a respirable emitted aerosol fraction and dose, (ii) presenting this to a small volume of medium that is representative of lung lining fluid, and (iii) measuring the low concentrations of drug released. We report developments in methodology for each of these steps and utilize mechanistic in silico modeling to evaluate the in vitro dissolution profiles in the context of plasma concentration–time profiles. The PreciseInhale aerosol delivery system was used to deliver Flixotide aerosol particles to Dissolv It apparatus for measurement of dissolution. Different media were used in the Dissolv It chamber to investigate their effect on dissolution profiles, these were (i) 1.5% poly(ethylene oxide) with 0.4% l -alphaphosphatidyl choline, (ii) Survanta, and (iii) a synthetic simulated lung lining fluid (SLF) based on human lung fluid composition. For fluticasone proprionate (FP) quantification, solid phase extraction was used for sample preparation with LC–MS/MS analysis to provide an assay that was fit for purpose with a limit of quantification for FP of 312 pg/mL |
|








